Overview
Cells stay alive by coordinating thousands of chemical reactions. Many of these reactions happen on cellular membranes – the thin, flexible surface that organize the cell’s behavior.
While we now understand individual proteins in great detail, and can observe the behavior of whole cells, the molecular events that happen on membranes – where proteins meet, move, and interact, have been difficult to see directly.
Our lab studies these membrane-based reactions by rebuilding them in simplified, controllable systems and watching them in real time using fluorescence microscopy.
Approach
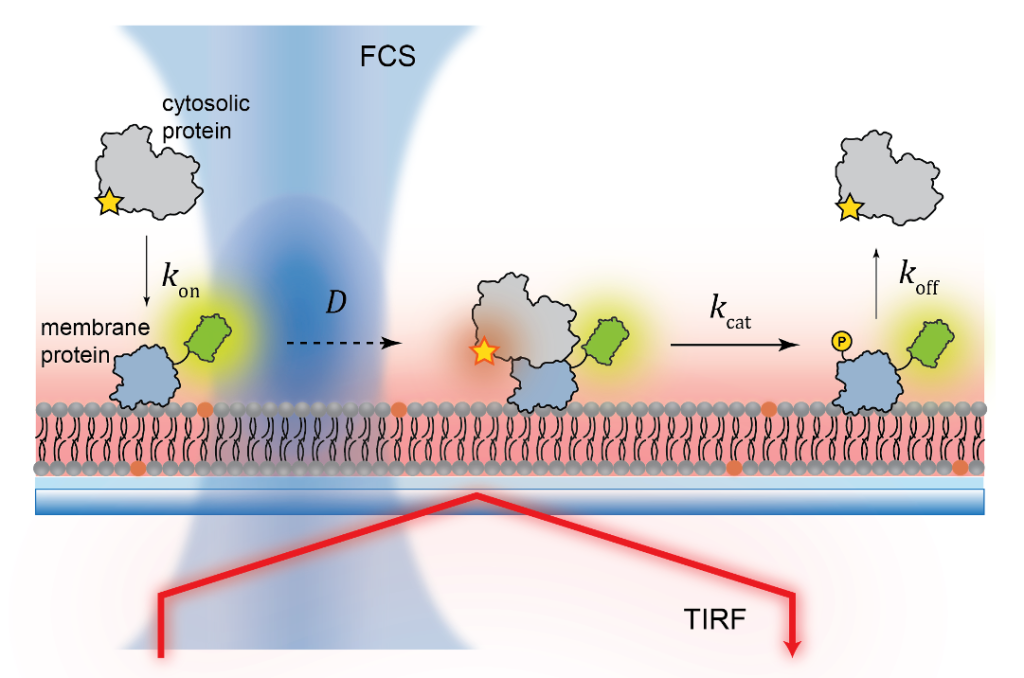
In vitro membrane reconstitution
- Model membranes (supported lipid bilayers, unilamellar vesicles, etc.) captures the phospholipid bilayer environment and are precisely controlled
- Readily compatible with quantitative optical methods
- Purified components are introduced to reconstitute the system of interest
- Allows precise measurement of key parameters and enables modeling
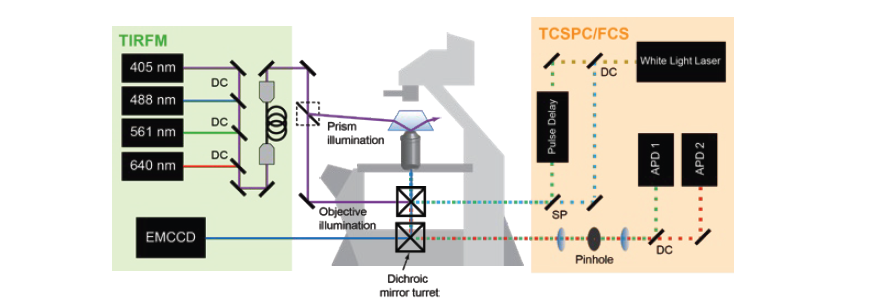
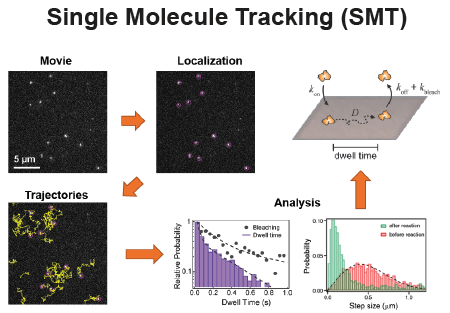
Fluorescence Spectroscopy and microscopy
- Fluorescence provides specificity and contrast required to resolve reactions on single bilayers.
- Total internal reflection fluorescence (TIRF) imaging tracks single-molecule diffusion and membrane interactions.
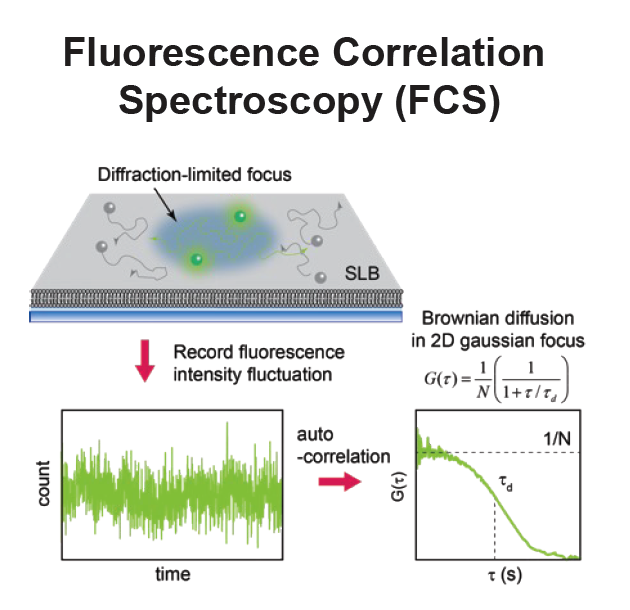
- Fluorescence correlation spectroscopy (FCS) provides surface density and lateral diffusion.
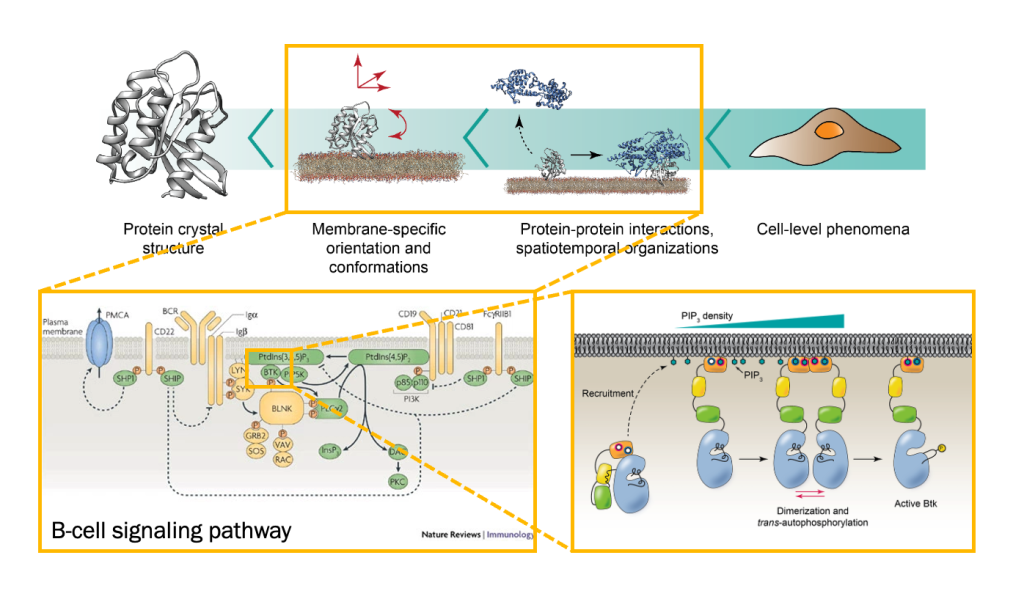
Project 1: the Role of Membrane Structure in Inflammation Pathway
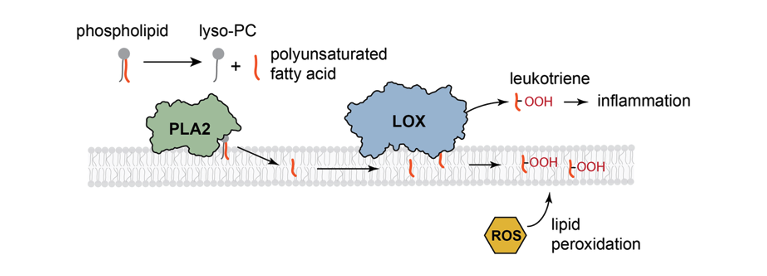
The inflammatory pathway is a key component of the immune response. One of its earliest steps occurs at the membrane, where the enzyme phospholipase A₂ (PLA₂) hydrolyzes phospholipids to release polyunsaturated fatty acids (PUFAs). These are subsequently oxidized by lipoxygenase (LOX) to generate leukotrienes, signaling molecules that initiate downstream inflammatory processes. In this project, our research focuses on how membrane structure influences the enzymatic activity governing these reactions.
- Jepson, T.A.; Hall, S.C.; Chung, J.K. “Single-molecule phospholipase A2 becomes processive on melittin-induced membrane deformations.” Biophys. J. 2022, 121(8), 1417–1423. [DOI]
- Min, S.; Picou, C.; Jeong, H.J.; Bower, A.; Jeong, K.; Chung, J.K. “Melittin–phospholipase A2 synergism is mediated by liquid–liquid miscibility phase transition in giant unilamellar vesicles.” Langmuir 2024, 40(14), 7456–7462. [DOI]
Project 2: Membrane Lipid Peroxidation
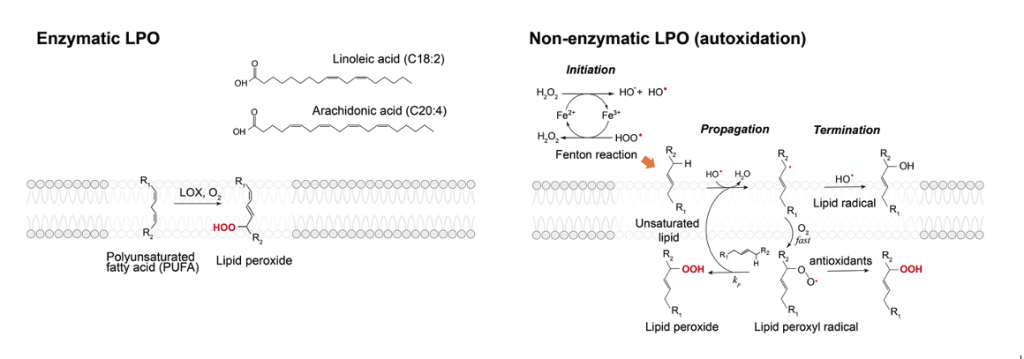
Lipid peroxidation (LPO) is a ubiquitous process associated with initiating and mediating the aging process, as it has been consistently demonstrated that there is an accumulation of lipid peroxides in older cell membranes. LPO is also associated with a legion of health concerns such as neurodegenerative diseases, cancer, cardiovascular diseases, and numerous others. Despite their fundamental role in aging and health, the mechanistic nature of LPO and its connection to the diseased state in cells remain elusive. This ambiguity stems from the fact that LPO studies are commonly carried out in live cell context, where quantitative measurements are difficult, or with lipids in organic solvents, where it lacks the membrane environment. This project focuses on the determination of chemical and physical mechanisms that underlie LPO using model membranes and quantitative fluorescence methods.
- Jeong, H.J.; Picou, C.; Jeong, K.; Chung, J.K. “Oxidation Kinetics of Fluorescent Membrane Lipid Peroxidation Indicators.” ACS Chem. Biol. 2024, 19, 1786–1793. [DOI]
- Jeong, H.J.; Min, S.; Villalon, L.; Jeong, K.; Chung, J.K. “Lipid-oxidative Enzymes and Fenton-like Reactions are Synergistic in Promoting Membrane Lipid Peroxidation.” JACS Au 2025, Accepted. [DOI]
Project 3: Protein Assembly on Membranes

Bruton’s tyrosine kinase (Btk) is a cytoplasmic tyrosine kinase that plays a central role in B cell signal activation and signal transduction. However, the active form of Btk requires localization to the plasma membrane – a process mediated by a lipid second messenger phosphatidylinositol (3-5)-triphosphate (PIP3). Upon binding PIP3 via the pleckstrin homology (PH) domain of the protein, Btk undergoes conformational changes, dimerization, and activation. Interestingly, while the PH domain facilitates dimerization and activation of the protein, other domains of Btk such as Src homology 3 (SH3) and proline-rich region (PRR) also facilitate the formation of 2-dimensional ordered structures much larger than protein dimers on the membrane, mediating a process known as biological phase separation. We hope to more fully understand the structures, material properties, and functional consequences of this process through the use of our reconstituted membrane systems and microscopy techniques.
Fenton, A.D.; Chung, J.K. “Membrane-dependent assembly of Bruton’s tyrosine kinase mediated by the Proline-rich region and SH3 domain.” Protein Science 2025, 34(8), e70213. [DOI]
